- Blog
- Security camera system
- Soshite watashi wa sensei ni
- Dashcam viewer not formatting
- Exped deepsleep mat 7-5 review
- Us open map 2021
- Arcane quest 3 editor
- Look deeper
- Umark 5 virus
- Disk inventory x upgrade
- Aiseesoft video converter ultimate freezes
- Tutorial animation desk
- Ecamm live to zoom
- Photo blender for windows
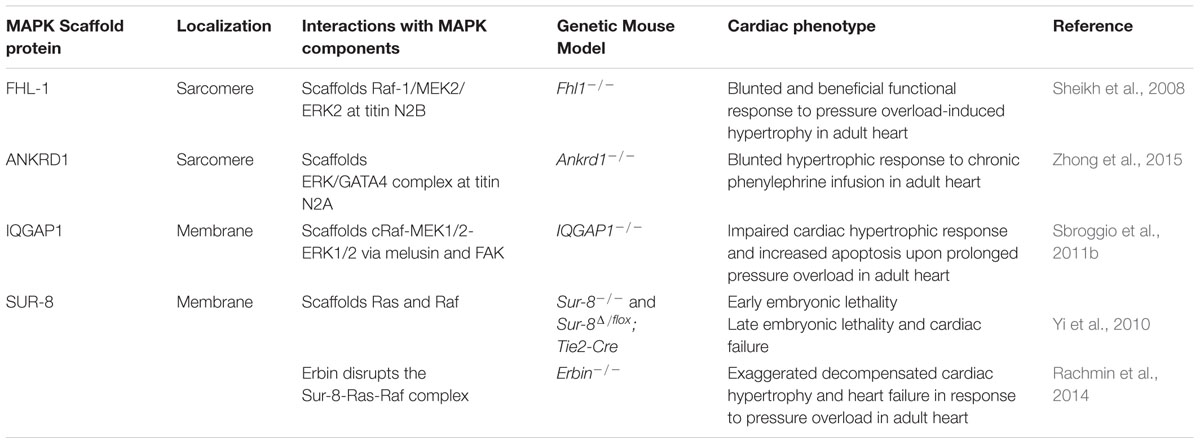
- Protein scaffold heart
- Ravensword shadowlands free download

The AG scaffolds were subsequently used for culturing neonatal rat cardiomyocytes, where high viability of the resulting cardiac constructs was observed under dynamic flow culture in a microfluidic bioreactor. The AG scaffolds also showed superior mechanical properties for the desired application and supported better adhesion, growth, and differentiation of myoblasts under static conditions. It consists of FAMs, MAPs and basal lamina proteins that confer structural integrity to the cardiac T-tubular membrane during contraction/relaxation cycles. Furthermore, the alginate/gelatin (AG) scaffolds better mimicked the native tissue in terms of interactions between components and protein secondary structure, and in terms of swelling behavior. These results indicate that the cardiac T-tubular system contains a subcellular scaffold closely resembling that of the costameres. We demonstrated that our scaffolds possessed highly porous and interconnected structures, and the chemical homogeneity of the natural ECM was well reproduced in both types of scaffolds.
Their morphological, physicochemical, and mechanical properties were determined and compared with those of natural porcine myocardium.

The readily reversible post-translational modification of phosphorylation is an essential regulatory mechanism in cardiomyocytes, facilitating these beat-to-beat adaptations. Scaffolds based on blends of a protein component, collagen or gelatin, with a polysaccharide component, alginate, were produced by freeze-drying and subsequent ionic and chemical crosslinking. The heart must rapidly respond to systemic changes to maintain an optimal supply of oxygen and nutrients to the body. Here, we propose the development of a class of protein/polysaccharide-based porous scaffolds for use as ECM substitutes in cardiac tissue engineering. The ECM hosts the cells and improves their survival, proliferation, and differentiation, enabling the formation of new tissue. One of the key elements for the success of tissue engineering is the use of a scaffold serving as artificial extracellular matrix (ECM). Tissue engineering has emerged as a viable approach to treat disease or repair damage in tissues and organs.
